# Magnetic Gas Lattice
[](https://stanslegacy.com/uploads/images/gallery/2024-10/4gxWQphJNL2SHlJ8-screenshot-from-2024-10-25-21-38-38.png)The forming **Argon ion** (Ar+) is now exposed to **Iron ions** (Fe+) (magnetic properties) experiencing and undergoing the same **Electron Extraction Process**.
Together, the **two** **ions** (Ar+/Fe+) form a covalent link up or **covalent bond** when the covalent electron of the **Argon ion** (Ar+) pair up and be shared with the valence electron of the **Iron ion** (Fe+).
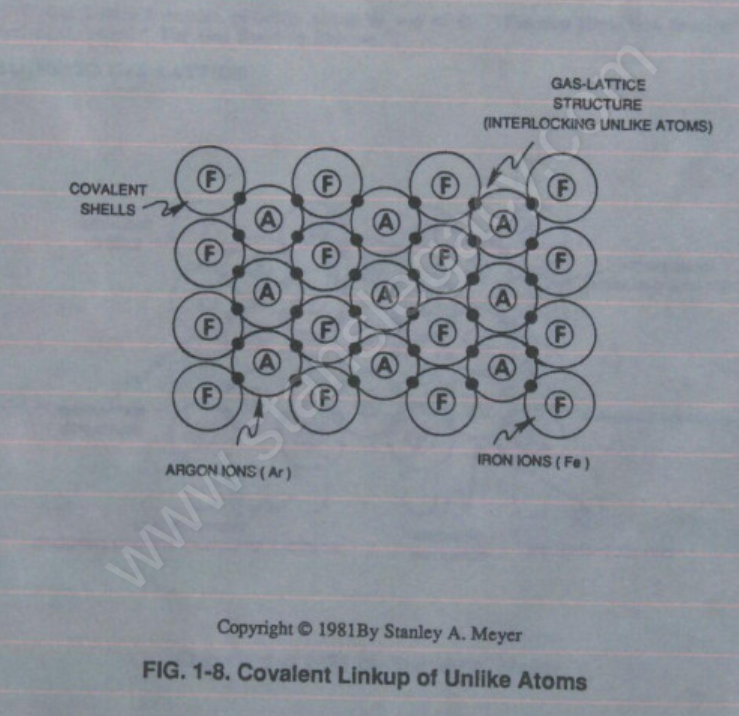
Covalent bonding of **Iron ions** (Fe+) to the **Argon ion** (Ar+) continues until a geometrical **Gas-Lattice Structure** is formed, as illustrated in Figure 1-8.
Stable-state of the **Gas-Lattice** occurs when the covalent shell of each unlike atom structure becomes full or filled up… the **Argon atom** (Ar) sees an covalent shell of 8 electrons while, at the same time, the **Iron atom** (Fe) sees an **covalent shell** (M shell) of 14 electrons.
Covalent bonding between like atoms does not occur due to the “stronger” **Electrical Attraction-Force** (qq') between the unlike atoms.
During Gas-Lattice formation, **Iron ions** (Fe+) can be replaced by other atoms exhibiting magnetic properties such as **Nickel ions** (Ni+) or **Cobalt ions** (Co+).
**Gas-Lattice formation** of unlike atoms by way of the **Electron Extraction Process** is, hereinafter, called **"The Gas Bonding Process"**.