The Fermi Level Barrier
![]() The Fermi level is a concept used in solid-state physics to describe the energy level at which electrons in a material have a 50% probability of being occupied. In the case of natural water and a 304L stainless steel electrode, the behavior of the Fermi level is important in understanding the electrochemical reactions that occur at the electrode-water interface.
The Fermi level is a concept used in solid-state physics to describe the energy level at which electrons in a material have a 50% probability of being occupied. In the case of natural water and a 304L stainless steel electrode, the behavior of the Fermi level is important in understanding the electrochemical reactions that occur at the electrode-water interface.
Natural water, such as that found in lakes, rivers, and oceans, is a complex mixture of dissolved gases, ions, and organic compounds. These components can interact with the electrons in the water and affect the position of the Fermi level. The Fermi level of natural water is generally close to the energy level of the water's highest occupied molecular orbital (HOMO), which is the energy level of the highest occupied electrons in the water molecule.
304L stainless steel is a commonly used material for electrodes in electrochemical applications due to its excellent corrosion resistance and durability. The Fermi level of the metal can be affected by several factors, including the presence of impurities, defects, or surface treatments. In the absence of any external influences, the Fermi level of 304L stainless steel is typically located within the energy band gap of the material.
When a 304L stainless steel electrode is submerged in natural water, electrochemical reactions can occur at the electrode-water interface. These reactions involve the transfer of electrons between the electrode and the water, which can cause changes in the position of the Fermi level. If the electrode is more electronegative than the water, electrons will flow from the water to the electrode, causing the Fermi level of the water to decrease and the Fermi level of the electrode to increase. This can result in the formation of an electric double layer at the electrode-water interface, which can affect the behavior of ions and molecules in the water.
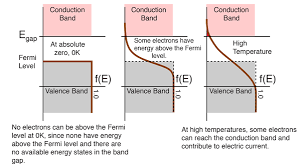 The behavior of the Fermi level of natural water and a 304L stainless steel electrode is important in many electrochemical applications, including corrosion prevention, electroplating, and water treatment. Understanding the electrochemical reactions that occur at the electrode-water interface can help to optimize these processes and improve their efficiency and effectiveness. The position of the Fermi level can be affected by several factors, including the composition of the water, the surface condition of the electrode, and the electrochemical reactions that occur at the electrode-water interface. By understanding these factors, researchers and engineers can develop new and improved methods for electrochemical applications.
The behavior of the Fermi level of natural water and a 304L stainless steel electrode is important in many electrochemical applications, including corrosion prevention, electroplating, and water treatment. Understanding the electrochemical reactions that occur at the electrode-water interface can help to optimize these processes and improve their efficiency and effectiveness. The position of the Fermi level can be affected by several factors, including the composition of the water, the surface condition of the electrode, and the electrochemical reactions that occur at the electrode-water interface. By understanding these factors, researchers and engineers can develop new and improved methods for electrochemical applications.
