Hydrogen Bonds
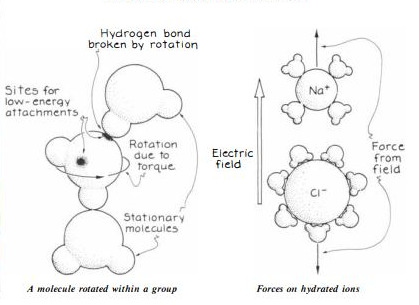
Hydrogen bonds are a type of intermolecular force that occurs when a hydrogen atom, covalently bonded to an electronegative atom, such as oxygen or nitrogen, interacts with a neighboring electronegative atom in another molecule. Hydrogen bonds are responsible for many important properties of substances, such as the high boiling point of water and the stability of protein and DNA structures.
To understand how hydrogen bonds form, it is important to first understand the basic structure of a water molecule. A water molecule consists of two hydrogen atoms covalently bonded to a single oxygen atom. Due to the electronegativity of the oxygen atom, the electrons in the covalent bonds are more attracted to the oxygen atom, creating a partial negative charge on the oxygen and a partial positive charge on the hydrogen atoms.
When two water molecules come close together, the partial positive charge of one hydrogen atom is attracted to the partial negative charge of the oxygen atom in the other molecule, forming a hydrogen bond. This interaction is not as strong as a covalent bond, but it is strong enough to cause water molecules to stick together, which is why water has a high surface tension and can form droplets.
Hydrogen bonds are also responsible for the unique properties of water, such as its high boiling point and heat capacity. When heat is added to water, the energy is used to break the hydrogen bonds between water molecules, rather than increasing the kinetic energy of the molecules. This means that water can absorb a lot of heat energy without changing temperature, making it an effective coolant.
Microwaves, on the other hand, heat water through a different mechanism. Microwaves are a form of electromagnetic radiation that can be absorbed by water molecules, causing them to vibrate rapidly and generate heat. This is why microwaves are effective at heating water-based substances, such as food or beverages.
In summary, hydrogen bonds are a type of intermolecular force that occurs when a hydrogen atom interacts with an electronegative atom in another molecule. They are responsible for many important properties of substances, such as the high boiling point of water and the stability of protein and DNA structures. Microwaves, on the other hand, heat water through the absorption of electromagnetic radiation, causing water molecules to vibrate and generate heat.